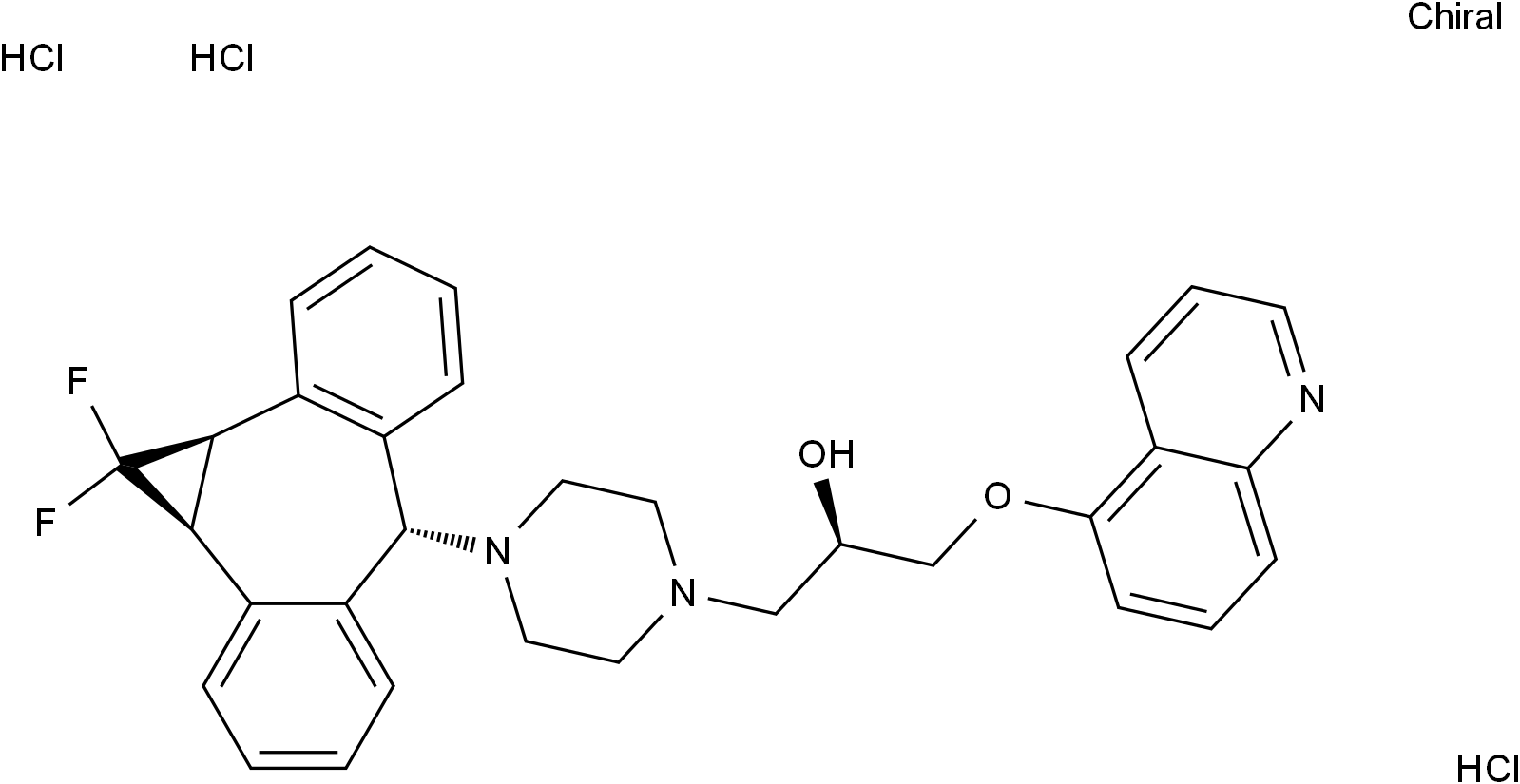
Zosuquidar trihydrochloride
CAS No. 167465-36-3
Zosuquidar trihydrochloride( LY-335979 trihydrochloride )
Catalog No. M12557 CAS No. 167465-36-3
A potent P-glycoprotein (P-gp) inhibitor with Ki of 60 nM.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 2MG | 36 | In Stock |


|
| 5MG | 58 | In Stock |


|
| 10MG | 81 | In Stock |


|
| 50MG | 340 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameZosuquidar trihydrochloride
-
NoteResearch use only, not for human use.
-
Brief DescriptionA potent P-glycoprotein (P-gp) inhibitor with Ki of 60 nM.
-
DescriptionA potent P-glycoprotein (P-gp) inhibitor with Ki of 60 nM; fully restores sensitivity to vinblastine, doxorubicin, etoposide, and Taxol in CEM/VLB100 cells at 0.1 uM; enhances the antitumor activity of Taxol in a MDR human non-small cell lung carcinoma nude mouse xenograft model.Blood Cancer Phase 3 Discontinued(In Vitro):Zosuquidar (0.3 μM; 48 h) enhances the cytotoxicity of DNR (substrates for P-glycoproteins) in P-glycoproteins active cell lines.Zosuquidar (5-16 μM; 72 h) treatment alone shows high cytotoxic concentration to drug-sensitive and MDR cell lines.(In Vivo):Zosuquidar (intraperitoneal injection; 30, 10, 3, or 1 mg/kg; once daily; 5 d) treatment shows a significant increase in life span.Zosuquidar (intraperitoneal injection; 30 mg/kg; once daily; 5 d) treatment shows the potentiation with a combined of Doxorubicin.
-
In VitroZosuquidar (0.3 μM; 48 h) enhances the cytotoxicity of DNR (substrates for P-glycoproteins) in P-glycoproteins active cell lines.Zosuquidar (5-16 μM; 72 h) treatment alone shows high cytotoxic concentration to drug-sensitive and MDR cell lines. ll Cytotoxicity Assay Cell Line:K562 and HL60 cells Concentration:0.3 μM Incubation Time:48 hours Result:Enhanced the cytotoxicity of DNR (substrates for P-glycoproteins) in K562/DOX cells more than 45.5-fold.Cell Cytotoxicity Assay Cell Line:CCRF-CEM, CEM/VLB100, P388, P388/ADR, MCF7, MCF7/ADR, 2780, 2780AD, UCLA-P3, UCLA-P3.003VLB cells Concentration:5-16 μM Incubation Time:72 hours Result:Showed IC50s of 6, 7, 15, 8, 7, 15, 11, 16, >5, >5 μM for CCRF-CEM, CEM/VLB100, P388, P388/ADR, MCF7, MCF7/ADR, 2780, 2780AD, UCLA-P3, UCLA-P3.003VLB cells, respectively.
-
In VivoZosuquidar (intraperitoneal injection; 30, 10, 3, or 1 mg/kg; once daily; 5 d) treatment shows a significant increase in life span.Zosuquidar (intraperitoneal injection; 30 mg/kg; once daily; 5 d) treatment shows the potentiation with a combined of Doxorubicin. Animal Model:Mice implanted with P388/ADR tumors Dosage:30, 10, 3, or 1 mg/kg Administration:Intraperitoneal injection; 30, 10, 3, or 1 mg/kg; once daily; 5 days Result:Exihibited a significantly increased survival compared to the group treated with Doxorubicin alone (P<0.001).Animal Model:Mice implanted with P388 or P388/ADR murine leukemia cells Dosage:30 mg/kg Administration:Intraperitoneal injection; 30 mg/kg; once daily; 5 days Result:Observed significant antitumor activity against the MDR P388/ADR cell lines when mice were treated with a combined dose of 30 mg/kg LY335979 and 1 mg/kg Doxorubicin (P=0.1).
-
SynonymsLY-335979 trihydrochloride
-
PathwayMembrane Transporter/Ion Channel
-
TargetP-glycoprotein
-
RecptorP-gp?(P-glycoprotein)
-
Research AreaCancer
-
IndicationBlood cancer
Chemical Information
-
CAS Number167465-36-3
-
Formula Weight636.9871
-
Molecular FormulaC32H34Cl3F2N3O2
-
Purity>98% (HPLC)
-
Solubility10 mM in DMSO
-
SMILESFC1([C@H]2[C@@H]1C3=C([C@@H](C4=CC=CC=C42)N5CCN(CC5)C[C@H](COC6=C7C=CC=NC7=CC=C6)O)C=CC=C3)F.Cl.Cl.Cl
-
Chemical Name1-Piperazineethanol, 4-[(1a.alpha.,6.alpha.,10b.alpha.)-1,1-difluoro-1,1a,6,10b-tetrahydrodibenzo[a,e]cyclopropa[c]cyclohepten-6-yl]-.alpha.-[(5-quinolinyloxy)methyl]-, hydrochloride (1:3), (.alpha.R)-
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Slate DL, et al. Anticancer Res. 1995 May-Jun;15(3):811-4.
2. Dantzig AH, et al. Cancer Res. 1996 Sep 15;56(18):4171-9.
3. Dantzig AH, et al. J Pharmacol Exp Ther. 1999 Aug;290(2):854-62.
molnova catalog



related products
-
Tariquidar methanesu...
A potent, specific P-gp inhibitor with Kd of 5.1 nM.
-
Zosuquidar trihydroc...
A potent P-glycoprotein (P-gp) inhibitor with Ki of 60 nM.
-
Dofequidar
A novel, orally active quinoline compound that can reverse P-glycoprotein (P-gp)-mediated MDR.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


